Channels
Special Offers & Promotions
New Liver Cancer Treatment Approved for Use in Europe
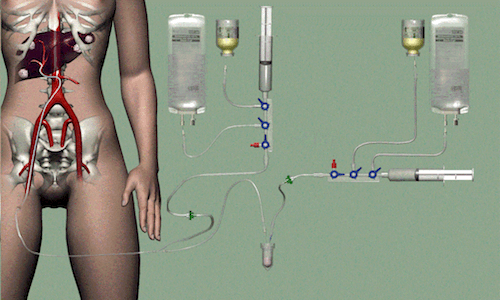
A new treatment for liver cancer developed by the University Medical Center (UMC) Utrecht has received the European CE mark for quality and safety.
This implies that hospitals throughout Europe can now start using this innovative treatment that uses radioactive holmium microspheres to attack liver tumors. The treatment is being marketed by Quirem Medical, a spin-off company of the UMC Utrecht.
It involves injecting radioactive beads into the hepatic artery, which then join the blood flow and become trapped in the tiniest blood vessels, located in and around the liver tumors. They therefore emit their radiation close to the tumor. This type of radiation treatment is also called radioembolization.
Patient-specific Treatment
"We consider our holmium microspheres as 'the next generation of microspheres,'" says Dr. Frank Nijsen, founder of Quirem. "Treating liver tumors with yttrium microspheres is already a proven and valued cancer therapy using radioembolization. The new holmium microspheres constitute the next step in the development of this technology. Because they show up on MRI scans and SPECT-CT, these microspheres can be tracked, allowing customized treatment for each individual patient. Now that we have been awarded CE marking, patients all over Europe can benefit."
The UMC Utrecht has been working for fifteen years to develop this innovative treatment modality. For the last six year, patients with liver tumors have been undergoing the treatment in a scientific setting. The spin-off company Quirem Medical was founded in 2013 to make the treatment available also to patients outside the UMC Utrecht. "The CE marking reflects our ambition to make our technology available to people around the world," Jan Sigger, CEO of Quirem Medical, points out. "We can now work to further develop this holmium treatment together with our clinical partners."
Other Organs
The research group at UMC Utrecht in charge of the project will work closely with Quirem Medical to adapt the new treatment for treating tumors in other organs as well. This year, research will start on the use of holmium microspheres to treat head and neck tumors. For that application, too, the unique image guidance with MRI introduces possibilities for closely monitoring the localized treatment of tumors and optimizing it as needed.
"I expect great things from this holmium therapy," says Prof. Maurice van den Bosch, an interventional radiologist at the UMC Utrecht. He was closely involved in the development of this new treatment. "Patients with liver tumors will benefit, and I hope that we will be able to treat patients with other tumors more effectively in the future as well."
Watch a video about this new treatment
Media Partners


