Channels
Special Offers & Promotions
New algorithm uncovers the secrets of cell factories
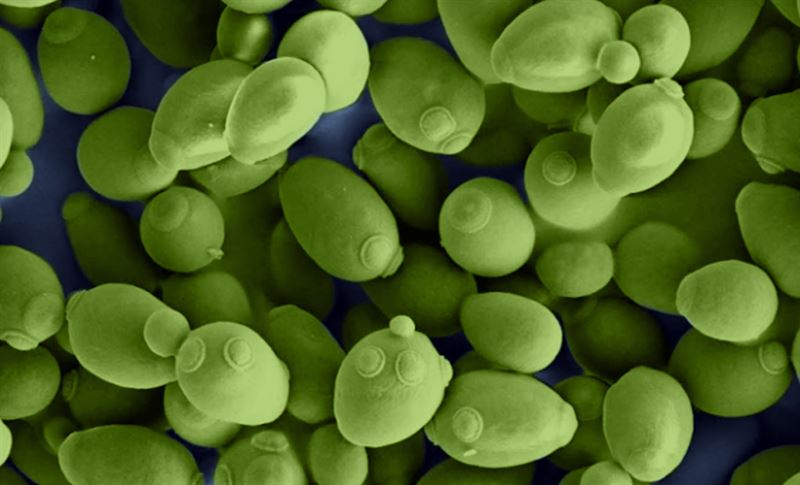
The researchers tested their model by simulating metabolism in more than 300 types of yeasts. When compared with measured, pre-existing knowledge, the researchers concluded that models with predicted kcat values could accurately simulate metabolism. The image shows common baker’s yeast, Saccharomyces cerevisiae
Drug molecules and biofuels can be made to order by living cell factories, where biological enzymes do the job. Now researchers at Chalmers University of Technology have developed a computer model that can predict how fast enzymes work, making it possible to find the most efficient living factories, as well as to study difficult diseases.
Enzymes are proteins found in all living cells. Their job is to act as catalysts that increase the rate of specific chemical reactions that take place in the cells. The enzymes thus play a crucial role in making life on earth work and can be compared to nature's small factories. They are also used in detergents, and to manufacture, among other things, sweeteners, dyes and medicines. The potential uses are almost endless, but are hindered by the fact that it is expensive and time consuming to study the enzymes.
“To study every natural enzyme with experiments in a laboratory would be impossible, they are simply too many. But with our algorithm, we can predict which enzymes are most promising just by looking at the sequence of amino acids they are made up of”, says Eduard Kerkhoven, researcher in systems biology at Chalmers University of Technology and the study's lead author.
Only the most promising enzymes need to be tested
The enzyme turnover number or kcat value, describes how fast and efficient an enzyme works and is essential for understanding a cell's metabolism. In the new study, Chalmers researchers have developed a computer model that can quickly calculate the kcat value. The only information needed is the order of the amino acids that build up the enzyme - something that is often widely available in open databases. After the model makes a first selection, only the most promising enzymes need to be tested in the lab.
Given the number of naturally occurring enzymes, the researchers believe that the new calculation model may be of great importance.
“We see many possible biotechnological applications. As an example, biofuels can be produced when enzymes break down biomass in a sustainable manufacturing process. The algorithm can also be used to study diseases in the metabolism, where mutations can lead to defects in how enzymes in the human body work”, says Eduard Kerkhoven.
More knowledge on enzyme production
More possible applications are more efficient production of products made from natural organisms, as opposed to industrial processes. Penicillin extracted from a mould is one such example, as well as the cancer drug taxol from yew and the sweetener stevia. They are typically produced in low amounts by natural organisms.
“The development and manufacture of new natural products can be greatly helped by knowledge of which enzymes can be used”, says Eduard Kerkhoven.
The calculation model can also point out the changes in kcat value that occur if enzymes mutate, and identify unwanted amino acids that can have a major impact on an enzyme's efficiency. The model can also predict whether the enzymes produce more than one "product".
“We can reveal if the enzymes have any ‘moonlighting’ activities and produce metabolites that are not desirable. It is useful in industries where you often want to manufacture a single pure product.”
The researchers tested their model by using 3 million kcat values to simulate metabolism in more than 300 types of yeasts. They created computer models of how fast the yeasts could grow or produce certain products, like ethanol. When compared with measured, pre-existing knowledge, the researchers concluded that models with predicted kcat values could accurately simulate metabolism.
The study Deep learning based kcat prediction enables improved enzyme constrained model reconstruction has been published in Nature Catalysis. The authors are Feiran Li, Le Yuan, Hongzhong Lu, Gang Li, Yu Chen, Martin Engqvist, Eduard Kerkhoven and Jens Nielsen. The researchers are active at Chalmers University of Technology.
About Chalmers University of Technology
Chalmers University of Technology in Gothenburg, Sweden, conducts research and education in technology and natural sciences at a high international level. The university has 3100 employees and 10,000 students, and offers education in engineering, science, shipping and architecture.
With scientific excellence as a basis, Chalmers promotes knowledge and technical solutions for a sustainable world. Through global commitment and entrepreneurship, we foster an innovative spirit, in close collaboration with wider society.The EU’s biggest research initiative – the Graphene Flagship – is coordinated by Chalmers. We are also leading the development of a Swedish quantum computer.
Chalmers was founded in 1829 and has the same motto today as it did then: Avancez – forward.
Media Partners


