Channels
Special Offers & Promotions
Deceptively Simple SCAPE 4D Microscope Brings Major Innovation to Live Cell and Whole Animal Imaging
Andor Zyla 5.5 sCMOS camera at the heart of new technique to perform real-time 3D imaging at unprecedented spatial resolution
 Advances in confocal, two-photon and light-sheet microscopy have rapidly advanced our understanding of cellular and subcellular structures and processes. However, live cell and whole animal imaging could be on the verge of even more breakthroughs following the publication in Nature Photonics of work by a team of Columbia University scientists using the fast and ultra-sensitive Andor Zyla sCMOS camera.
Advances in confocal, two-photon and light-sheet microscopy have rapidly advanced our understanding of cellular and subcellular structures and processes. However, live cell and whole animal imaging could be on the verge of even more breakthroughs following the publication in Nature Photonics of work by a team of Columbia University scientists using the fast and ultra-sensitive Andor Zyla sCMOS camera.
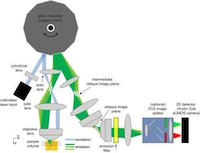
Led by Elizabeth Hillman, associate professor of biomedical engineering at Columbia University Medical Center (CUMC), and her graduate student, Matthew Bouchard, the team has successfully developed the SCAPE (Swept Confocally Aligned Planar Excitation) 3D microscope, which eliminates the need to mount samples or other special preparation and is capable of imaging freely moving living samples in real-time at speeds 10 to 100 times faster than current laser-scanning microscopes.
According to Professor Hillman, “Unlike conventional light-sheet microscopes that utilize a pair of cumbersomely-positioned objective lenses, SCAPE uses a single-objective lens with a light sheet sweeping across the field of view to capture 3D images without moving the sample or the objective. This combination makes SCAPE extremely fast, versatile and simple to use, as well as surprisingly inexpensive, and could be transformative in bringing the ability to capture high-speed 3D cellular activity to a wide range of living samples. With the Andor Zyla sCMOS camera set to read out 2,560 x 80 micron images at 2,404 frames per second we have demonstrated the ability of SCAPE to image living organisms, including Drosophila melanogaster larvae and zebrafish at up to 48 volumes per second.
"SCAPE has also captured neurons flashing in a living mouse brain and, unlike other 3D imaging systems, doesn't need to constantly readjust either the imaging objective lens or the sample, allowing the subject to move freely throughout the imaging process. The ability to perform real-time 3D imaging at cellular resolution in behaving organisms is a new frontier for biomedical and neuroscience research," said Professor Hillman. "With SCAPE, we can now image complex, living things, such as neurons firing in the rodent brain, crawling fruit fly larvae, and single cells in the zebrafish heart while the heart is actually beating spontaneously – this has not been possible until now."
Many further applications for SCAPE are envisaged by the CUMC researchers, including capturing images of cellular replication, function, and motion in living tissue, and imaging 3D cell cultures, as well as capturing 3D images of dynamics in microfluidic and flow-cell cytometry structures. These areas may be particularly enhanced by SCAPE as these are applications where molecular biology imaging methods have fallen behind the latest tools and techniques.
Unlike previous CMOS or CCD technologies, the Andor Zyla models set radical new benchmarks in their unique ability to deliver simultaneously highest specifications in sensitivity, resolution, speed, dynamic range and field-of-view. The camera is based around a large 4.2 or 5.5 megapixel sensor with 6.5 micron pixels and is capable of 100 fps sustained (via Camera Link; 40 fps via USB3), making it ideal for applications in cell microscopy, astronomy, digital pathology, and high content screening. The Rolling and Global shutter exposure modes further enhances application flexibility. Global shutter in particular offers an ideal means to simply and efficiently synchronize the Zyla with other ‘moving’ devices, such as stages or light switching sources, and eliminating the possibility of spatial distortion when imaging fast-moving objects.
Andor's microscopy systems and scientific cameras address a broad range of optical microscopy techniques including laser spinning disk confocal microscopy, photo-bleaching, activation, conversion and ablation, TIRF, white light spinning disk confocal, calcium ratio imaging, comet assay and bioluminescence.
Media Partners


