Channels
Special Offers & Promotions
Haematology biomarkers for COVID-19 assessment discussed in new HORIBA Medical White Paper
How routinely measured biological markers could contribute to the monitoring of Coronavirus (COVID-19)
As experts in haematology HORIBA UK Ltd, Medical announces that the Company has published a white paper entitled, ‘COVID-19 screening, prognosis and severity assessment with biomarkers for management of patients’ [1]. Written in collaboration with academic partners, the paper provides a review of routinely measured biomarkers that can provide additional information to help in the assessment of the severity of COVID-19 infection; therefore, supporting clinical decision making for risk stratification and patient management.
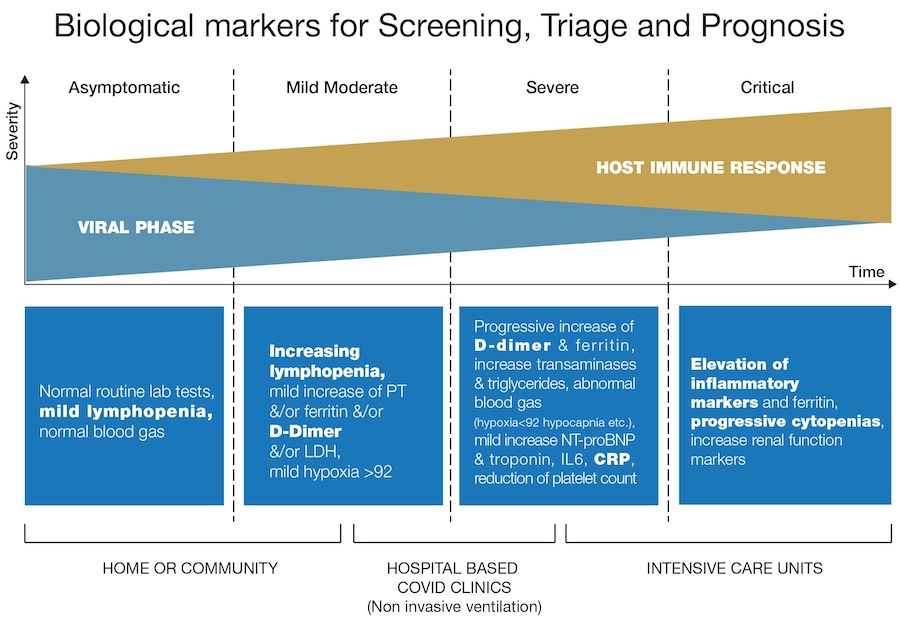
The gold standard for confirmation of COVID-19 diagnosis is through the detection by molecular analysis of the viral genome using qPCR. However, detection of the virus is not a predictor of the severity of the disease, so the use of a comprehensive panel of easily measured biological markers with clinical observations can support screening and prompt patient management decisions.
Several biomarkers have been observed to be abnormal in COVID-19 infected patients and the relevance of identifying them lies in decreasing the possibility of misdiagnosing severe COVID-19 and providing more insightful information for better management of COVID-19 patients. Many cohorts of different populations have been reported, principally from China, showing abnormal laboratory assessments of complete blood count, liver and renal function, biochemical and coagulation testing and inflammatory factors, amongst others.
The biological markers that can support COVID-19 screening, triage and prognosis discussed in the white paper can all be measured on routine haematology analysers. These markers are white blood cells (WBC), platelets, D-dimer, C-Reactive Protein (CRP) and fibrinogen; the paper provides a literature review and overview of the response of each to COVID-19 clinical severity. For example, lymphopenia and increases in CRP plus cytokines, D-dimer and fibrinogen have all been noted in a number of scientific papers to correlate with COVID-19 disease severity.
All these markers could aid in triage to help better identify COVID-19, particularly those patients most likely to require intervention and/or intensive care. They can also facilitate the continuation of other critical clinical services by aiding the separation of COVID-19 patients. Where possible, they can even be used for testing in the community to avoid the unnecessary travel of vulnerable or possibly infected individuals.
Supporting the new white paper, HORIBA Medical has also published a series of three Focus Notes which review ‘How biological markers could contribute to the monitoring of COVID-19?’ [2]. Each focuses on a different panel of biomarkers, with the first discussing haematology biological markers, the second highlighting the inflammatory marker CRP and the third detailing the use of haemostasis markers. These, as well as the white paper, are all available for download at HORIBA Medical’s Coronavirus (COVID-19) dedicated web page.
As experts in haematology, HORIBA Medical designs and manufactures a broad portfolio of systems capable of measuring a range of potential COVID-19 biomarkers, both at the point-of-care and in the laboratory. For example, in addition to Full Blood Count with WBC differentials, lymphocytes and platelets available on all of its haematology analysers, HORIBA combines these parameters with CRP on its Microsemi CRP and Pentra MS CRP analysers. The Microsemi CRP is unique as it can deliver these parameters at the point-of-care to support rapid triage and clinical decisions. Completing the portfolio, HORIBA’s Yumizen G range of coagulation analysers provide accurate measurement of fibrinogen and D-dimer.
References
- Siatka, C., Eveillard, M., Nishimura, J., Duroux, C., Ferrandi, G. (2020). COVID-19 screening, prognosis and severity assessment with biomarkers for management of patients. HORIBA Medical White Paper. Published April 2020. https://static.horiba.com/fileadmin/Horiba/Products/Medical/By_Segment/COVID-19_screening__prognosis_and_severity_assessment_with_biomarkers_HORIBA_Medical.pdf (Accessed June 2020)
- HORIBA Medical (2020). How biological markers could contribute to the monitoring of COVID-19? Focus Note #1: Hematology Biological Markers. https://www.horiba.com/en_en/products/by-segment/medical/hematology-analyzer/coronavirus-covid19/how-biological-markers-could-contribute-to-the-monitoring-of-covid-19-focus-note-1-hematology-biological-markers/ (Accessed June 2020)
- HORIBA Medical Coronavirus (COVID-19) dedicated web page. www.horiba.com/en_en/products/by-segment/medical/hematology-analyzer/coronavirus-covid19/ (Accessed June 2020)
About HORIBA UK Ltd
Resulting from over 30 years of understanding and meeting its customers’ individual needs, the HORIBA UK Ltd Medical team holds a wealth of expertise and is a well-respected and renowned specialist in the design, manufacture and support of automated in vitro diagnostic systems.
HORIBA Medical facilitates the entire analytical process, both in the laboratory and at the point of care, from process and analyser optimisation, analysing samples and managing data to remote monitoring and e-Learning. Furthermore, in addition to NHS and private healthcare settings, HORIBA Medical products are proving their worth in many other areas, including: sports science, food and beverage, pharma and nutraceuticals, and clinical research.
HORIBA Medical’s product and service combinations all deliver quality results, flexibility, security and process efficiency. These merits in turn ensure effective clinical decision making, enhanced patient care pathways and improved patient outcomes.
Media Partners