Channels
Special Offers & Promotions
Novozymes Biopharma US Inc
Products
Contact Novozymes Biopharma US Inc
All articles from Novozymes Biopharma US Inc
Novozymes expands collaboration with Upperton Limited to offer customers new albumin conjugation solution
Jan 26, 2012
Novozymes Biopharma Presents Research on Recombinant Human Albumin at 2011 PepTalk Conference
Jan 9, 2011
Novozymes spotlights innovative solutions for medical devices, drug delivery and formulation at CPhI 2010
Sep 10, 2010
Novozymes showcases leading biopharmaceutical solutions at BioProcess International 2010
Sep 3, 2010
SAFC and Novozymes Biopharma extend joint agreement for the exclusive distribution of LONGR3 growth factor
Aug 31, 2010
Media Partners


 Novozymes Biopharma, part of Novozymes A/S, a world leader in bioinnovation, has announced that it has expanded its partnership with Nottingham-based research and development company, Upperton Limited, to offer customers a new albumin conjugation solution. By linking drugs to Novozymes' new Recombumin Flex recombinant human albumin (rAlbumin), using conjugation, their pharmacokinetic and pharmacodynamic properties can be dramatically enhanced. As a result...
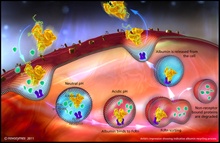
Novozymes Biopharma, part of Novozymes A/S, a world leader in bioinnovation, has announced that it has expanded its partnership with Nottingham-based research and development company, Upperton Limited, to offer customers a new albumin conjugation solution. By linking drugs to Novozymes' new Recombumin Flex recombinant human albumin (rAlbumin), using conjugation, their pharmacokinetic and pharmacodynamic properties can be dramatically enhanced. As a result...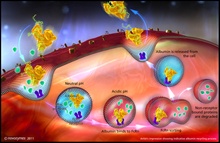 Novozymes Biopharma, part of Novozymes A/S, the world leader in bioinnovation, has unveiled its enhanced next-generation albumin technology, which was developed in collaboration with the University of Oslo, Norway, one of the world's leading institutions in the research of albumin variants and the neonatal Fc receptor (FcRn). Built on Novozymes' original Albufuse platform, the proprietary Albufuse Flex technology has been designed to enable users to adapt and control the pharmacokinetics of their target protein or peptide with retained efficacy, ensuring flexibility and optimal use...
Novozymes Biopharma, part of Novozymes A/S, the world leader in bioinnovation, has unveiled its enhanced next-generation albumin technology, which was developed in collaboration with the University of Oslo, Norway, one of the world's leading institutions in the research of albumin variants and the neonatal Fc receptor (FcRn). Built on Novozymes' original Albufuse platform, the proprietary Albufuse Flex technology has been designed to enable users to adapt and control the pharmacokinetics of their target protein or peptide with retained efficacy, ensuring flexibility and optimal use... Novozymes Biopharma, part of Novozymes A/S world leader in bioinnovation, is presenting two scientific posters on the manufacturing and use of recombinant albumin in formulation during PepTalk 2011, 10-14 January, San Diego, US. The posters cover the company's latest research in the use of recombinant human albumin and its ability to address many of the formulation challenges faced by the industry
Novozymes Biopharma, part of Novozymes A/S world leader in bioinnovation, is presenting two scientific posters on the manufacturing and use of recombinant albumin in formulation during PepTalk 2011, 10-14 January, San Diego, US. The posters cover the company's latest research in the use of recombinant human albumin and its ability to address many of the formulation challenges faced by the industry