Channels
Special Offers & Promotions
EKF Diagnostics Publishes
Educational guide available for free download from EKF’s new Diabetes Portal
EKF Diagnostics, the global in vitro diagnostics company, has published a guide to ‘Diabetes and HbA1c testing’ which can be found at EKF’s new Diabetes Portal .This new educational guide draws on EKF’s expertise in the diagnosis and monitoring of diabetes and associated conditions. It provides an overview of the global diabetes ‘epidemic’, symptoms and complications, through to discussion on methods for diagnosis and monitoring using both glucose and HbA1c testing, with consideration given to factors influencing their measurement.
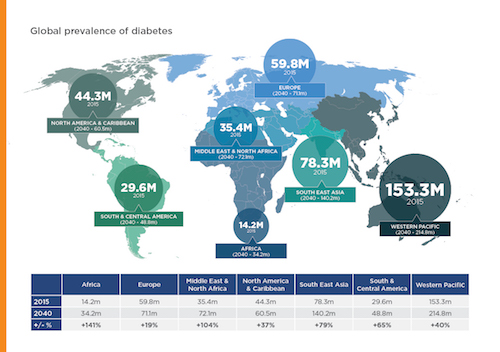
Diabetes is a growing issue, particularly in developing countries, with five million people dying from diabetes related complications in 2015 alone. Although not an ‘epidemic’ in the conventional sense, there are currently 415 million people living with diabetes and this is predicted to grow to 642 million by 2040. Approximately 46% of people living with diabetes are doing so without a full and proper diagnosis with subsequent complications, and associated healthcare costs [1].
There are multiple options for the diagnosis of diabetes, most of which involve measuring the level of glycemic control a person exhibits. In addition to methods such as fasting plasma glucose and two hour plasma glucose, another option is to use glycated hemoglobin (HbA1c) which reflects average plasma glucose over an 8-12 week period. As well as lab-based testing, WHO has approved HbA1c for diabetes diagnosis with a Point of-Care-Testing (POCT) device, providing the test is undertaken by a trained professional adhering to an appropriate External Quality Assurance scheme and using a methodology traceable to the IFCC reference method [2]. POCT HbA1c testing gives a strong indication of both diabetes and pre-diabetes within a timeframe that enables immediate intervention.
Since it is not impacted by the same issues as blood glucose monitoring, HbA1c testing is fast becoming the preferred technique for diabetes and pre-diabetes diagnosis and monitoring. There are situations where use of HbA1c is not appropriate though. EKF’s new Guide includes discussion on both glucose and HbA1c testing and where factors may influence measurement of both diagnostic markers.
“The often ‘low to no’ maintenance approach to disease management of diabetes is not only dangerous but can also significantly contribute to ongoing healthcare costs. At present, approximately 12% of global health expenditure is spent on diabetes, and without significant changes to the way patients and health systems monitor glycemic control this will surely rise,” said Gavin Jones, Global Product Manager for Diabetes Care at EKF Diagnostics. “To enhance patient outcomes and reduce the cost for long-term healthcare of the diabetic and pre-diabetic population, we aim to improve patient access to diabetes care techniques, whatever their location. This can be achieved by providing affordable, easy-to-use POCT analyzers and chemistry assays for both diabetes diagnosis and monitoring.”
EKF’s expertise and product range cover all aspects of diabetes care, from research and hospital laboratories to diabetes clinics, emergency rooms and GP surgeries. Products include the Biosen C-Line glucose analyzer, which uses chip sensor technology to provide low cost, fast and lab accurate glucose results. Quo-Test® and Quo-Lab® point-of-care HbA1c analyzers that deliver results meeting NGSP and IFCC POC requirements in four minutes. And lastly, to aid the diagnosis and monitoring of diabetes related conditions such as ketoacidosis, EKF offers Beta-Hydroxybutyrate LiquiColor® Reagent and the handheld STAT-Site® M β-HB strip-based analyzer.
Download the new Guide to guide to ‘Diabetes and HbA1c testing'
Media Partners


