Channels
Special Offers & Promotions
Residual Solvents analysis of cannabinoid products by Headspace GC-MS

Introduction
Hemp and marijuana are becoming more popular because of legalisation in multiple countries. The increase of this market also increases the offer of products and therefore the need to monitor different components.
This application focusses on Residual Solvent analysis but Scion Instruments has an extended portfolio including potency analyses (AN091, 92, 93 and 130) and Terpene analysis (AN133 and 136) Residual Solvents are organic volatile products that are often used for the extraction of CBD and THC from marijuana leaves. Unfortunately these Residual Solvents can cause health risks when after extraction these components are not removed accurately. Therefore it is important to monitor these components to asses the quality and safety of the product for safe consumption.
Figure 1 shows the SCION Instruments 436 GC with single quad (SQ) mass spectrometer together with the Scion HT3 Headspace sampler. On which this method is applicable to.

Figure 1. SCION HT3 Headspace sampler together with the SCION Instruments 436 GC with SQ-MS.
Experimental
This analysis can be implemented on the 436-GC and the 456-GC platform. The analysis was performed on the SCION Instruments 456-GC analyser equipped with SQ MS and SCION Instruments HT3 Headspace autosampler (Table 2). There are multiple Residual Solvent components known but for this application the focus is on the 20 most common solvents (Table 1).
Table 1: Residual Solvent components
| Nr. | Component | Nr. | Component |
| 1 | Methanol | 11 | Chloroform |
| 2 | Pentane | 12 | Cyclohexane |
| 3 | Ethanol | 13 | Benzane |
| 4 | Ethyl ether | 14 | 1,2 -Dichloroethane |
| 5 | Acetone | 15 | Heptane |
| 6 | 2-Propanol | 16 | Trichloroethylene |
| 7 | Acetonitrile | 17 | Toluene |
| 8 | Methylene chloride | 18 | p-Xylene |
| 9 | Hexane | 19 | m-Xylene |
| 10 | Ethyl Acetate | 20 | o-Xylene |
The focus of the calibration lies on these 20 components, the calibration curves for the Residual solvent standards were prepared between 0.5 and 25 µg/ml. The Quality Control (QC) sample was made from 7.5 µg/ml for all components. When looking at the sample preparation it is shown that a different approach is necessary depending on sample matrix. Keep in mind that hemp oil and capsules contain the same product but the sample preparation will be different.
In this assessment the choice was made to perform the analysis as an external calibration. When an internal standard is preferred α,α,α trifluorotoluene is recommended. To all samples 1 ml of saturated Sodium Sulphate solution (salting out) has to be added. This was done in order to increase sensitivity and headspace efficiency.
Table 2. Instrumentation operating conditions.
| Injector | Splitless 150:1, 200 °C |
| Column | 624-MS |
| Oven Program | 35°C (1.0 min), 15°C/min to 180°C (0 min), 30°C/min to 240°C (0.5 min) |
| Carrier | Helium, 1.6 ml/min |
| Autosampler | SCION HT3 |
| Software | MS work station/ HT3 Teklink |
| Oven temperature | 80°C |
| Transfer line temperature | 140°C |
| Sample temperature | 80°C |
| Sample equilibrium time | 12 min |
| MS transfer line temperature | 250°C |
| Ion source temperature | 200°C |
| Ionization mode | EI |
| Scan start | 1.25 |
| Scan mode | Full Scan |
Some components like methanol have a high partition coefficient (K), this means the compound has better affinity to the sample matrix instead of the gas phase. In order to lower the K value, matrix modification techniques have to be used. Salting out is one of the most common techniques, but also changing the phase ratio can decrease K and increase sensitivity. Figure 2 shows the effect of the addition of the saturated salt.
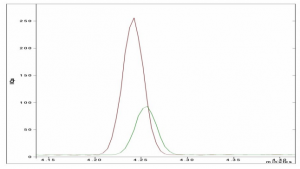
Figure 2: The response of Cyclohexane with (red) and without (green) the addition of salt.
Results
The precision of the method was obtained by seven consecutive injections containing 7.5 µg/ml of each component. It was shown that the RSD for the components all lie below 7.0 %. which is really good for this method. The repeatability of each component can be found in table 3. All the chromatographic peaks were identified with the NIST library. Figure 4 shows examples of these target peaks from a 5 µg/ml standard containing a mix of all the components.
The calibration curves for the Residual Solvents standards were prepared between 0.5 to 25 µg/ml. Most components had a correlation coefficient (R2) of 0.999 or greater which is perfect for this application. Figure 3 shows an example of the calibration curve for three components.
From the linearity the limit of detection (LOD) and limit of quantitation (LOQ) were calculated.
The QC sample was analysed using seven injections, the average concentrations are shown in table 3 together with the LOD and LOQ. After analysis of the hemp oil it showed that the sample contained 2– propanol and traces of both methanol and pentane, the rest of the components were not detected. The concentration of these components are also shown in table 3.
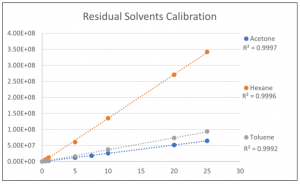
Figure 3: Three example calibration curves of the Residual Solvents standard.
Table 3. Results of different Residual Solvents components
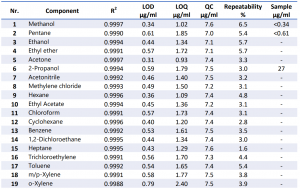
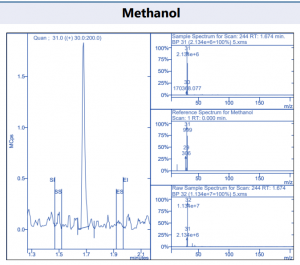
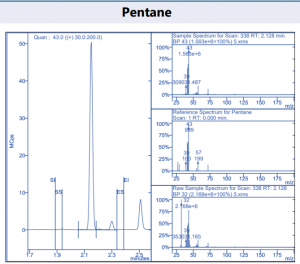
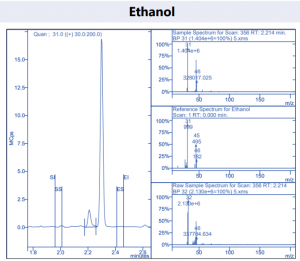
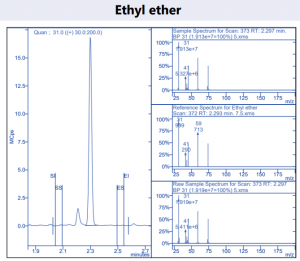
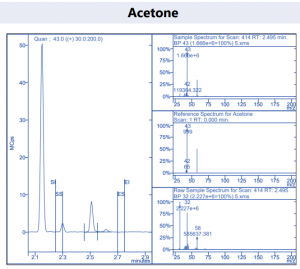
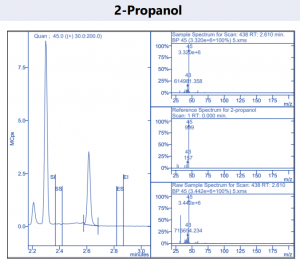
Figure 4. Target compound example peaks.
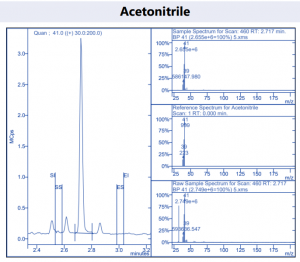
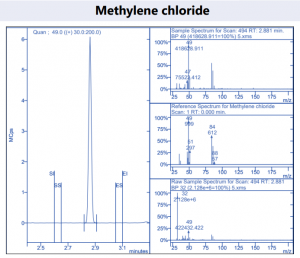
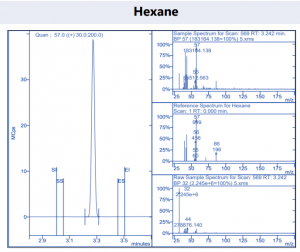
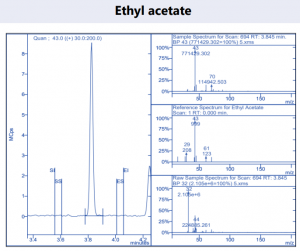
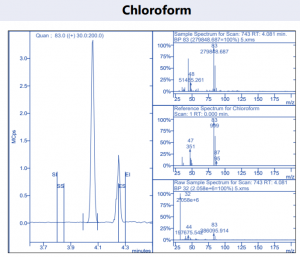
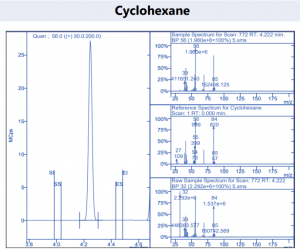
Figure 4. Target compound example peaks part 2.
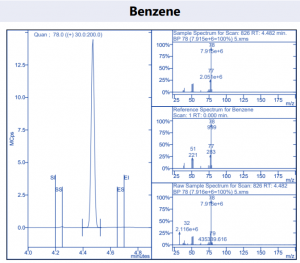
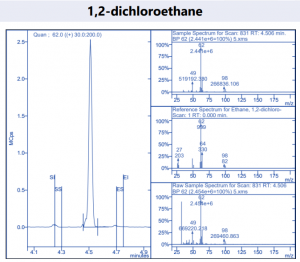
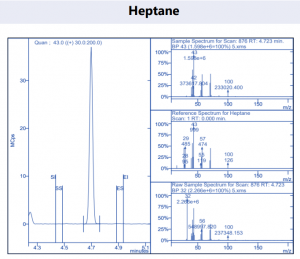
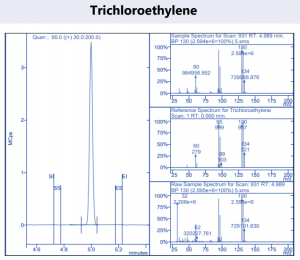
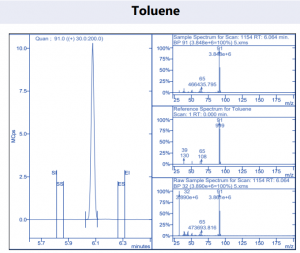
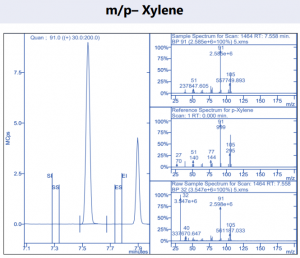
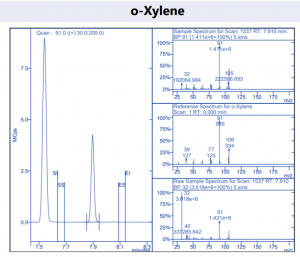
Figure 4. Target compound example peaks part 3.
Conclusion
The SCION 4X6-GC analyser equipped with a split/spitless injector, Scion Instruments column, SQ-MS and HT3 Headspace Sampler is capable of analysing Residual Solvents from cannabis products in a qualitative and quantitative way. It is also possible to perform this application with the SCION Instruments Versa Automated Headspace Vial Sampler. The method developed is well suited for the analysis of residual solvents. Experimentation showed that the addition of saturated salt is important to create sensitivity in the low concentration area.
Download Application Note
Download complete Application Note: Residual Solvents analysis of cannabinoid products by Headspace GC-MS
SCION Instruments Products
Find out more about the HT3 Static and Dynamic Headspace System and the SCION GC. If you would like to talk to a member of our sales team about any of these products, please feel free to contact us and someone will be happy to help.
Media Partners


